Obtaining the accurate three-dimensional (3D) structure information of the target is helpful for the discovery and optimization of drug candidates, as well as the mechanism of action (MOA) studies. As a leading service provider in the field of structural biology, relying on the first-class structural biology technology platform that covers X-ray crystallography, cryo-electron microscopy (Cryo-EM) technology and protein NMR spectroscopy, Creative Biostructure provides a comprehensive solution of target structure determination, called MagHelix™ Gene-to-Protein and Gene-to-Structure, complemented with the most advanced biophysical methods to characterize target and target-ligand interaction. Our services support structure-guided lead discovery and optimization, as well as biological drug discovery, such as therapeutic antibody discovery.

Our molecular cloning service includes the design and preparation of expression constructs. We provide customers with all commercially available vectors for construct design. Various purification tags are supplied for constructing recombinant plasmids, such as SUMO, MBP, GST, His tags, etc. We also have various inserts such as GPCR, kinases, phosphatases, and epigenetic targets. In this process, our scientists will determine the appropriate construct boundaries according to experience, precedent, and specific research applications and propose the optimal expression strategy. Our experienced technicians are able to perform site mutation, deletion, and multi-parallel construct design equipped with advanced techniques.
The service includes recombinant protein expression testing, optimization of protein expression and purification, as well as large-scale protein overexpression and chromatographic purification. Creative Biostructure provides customers with high-performance protein expression systems including E. coli, yeast, insect cells, and mammalian cells. We purify the target protein using the AKTA FPLC systems with multiple chromatographic methods (our standard protocol includes affinity chromatography, ion-exchange chromatography, and size-exclusion chromatography). After purification, the protein can be tested with UV spectrophotometry, SDS-PAGE, and western blot to ensure > 95% purity. The service is available as a stand-alone one or in combination with downstream applications. It is worth mentioning that we support custom membrane protein production.
X-ray crystallography has always been one of the mainstream techniques to solve the high-resolution structure of drug targets. In addition, it provides reliable structural information on target-ligand interaction sites and binding patterns during the screening of hits and optimization of lead compounds. Crystallographers from Creative Biostructure can solve the crystal structure of individual proteins, protein-ligand complexes, and biological complexes. Our crystallography science team expertise includes detailed target know-how, especially in membrane proteins, kinases, phosphatases, and epigenetic proteins.
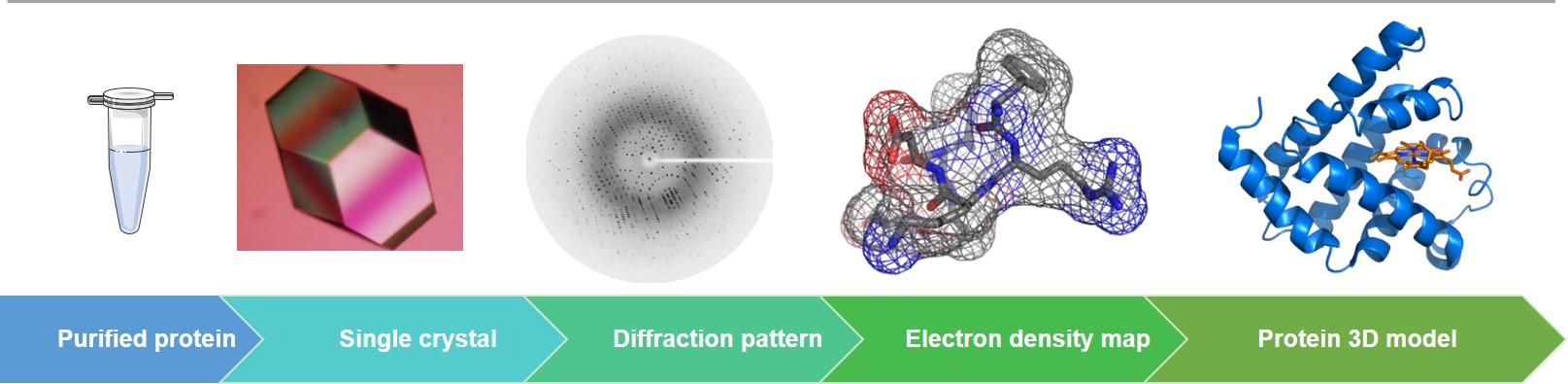
Before crystallization, the monodispersity of the target protein can be assessed using dynamic light scattering (DLS). With high-throughput crystallization equipment and various crystallization methods (such as vapor diffusion crystallization, co-crystallization, and seeding), crystallization conditions can be initially screened out. To obtain high-quality crystals that meet the requirements of X-ray diffraction, we will design the optimal optimization strategy and several crystallization conditions will be further optimized, such as protein concentration, pH value, salts, precipitants, additives, detergents, ligands, temperature, and so on; secondly, different crystal growing methods will be evaluated, e.g., seeding, cross seeding and crystal re-growth techniques. During the structure-guided lead discovery and optimization, the target protein can be co-crystallized with compounds.
X-ray diffraction data is collected on synchrotron beamlines to ensure the highest resolution for your project (providing in-house Rigaku X-ray equipment if necessary). Our data collection specialists can improve the diffraction quality by using cryoprotectants, annealing agents, and dehydrants. After collecting the X-ray data, our scientists find out the initial phases by using the molecular replacement (MR) or single-wavelength anomalous dispersion (SAD) and multi-wavelength anomalous dispersion (MAD) methods. And then, we will build the structural model and refine it. The quality of the final model will be validated through programs such as PROCHECK. Upon request, we can also assist customers in the detailed analysis of the individual protein structure or co-crystal structure to facilitate structure-based drug design.
We can prepare crystal-grade protein samples from scratch or obtain them from customers. Moreover, Creative Biostructure also owns the off-the-shelf crystal structure service if customers need it. In-house developed high-purity proteins and crystal structures are readily available for compound soaking and co-crystallization.
Over the past few years, as the hardware and software involved in cryo-EM have improved, we can utilize cryo-EM single-particle analysis (SPA) technology to provide the required target structure information for structure-based drug design. This technology makes it more and more routine to analyze the 3D structure of biomacromolecule complexes.

Creative Biostructure's cryo-EM SPA service covers the entire technical workflow. The target protein sample for analysis can be provided by the customer or customized by us, followed by negative staining EM. Negative staining EM is a convenient method for screening homogeneous samples and provides an initial model for obtaining high-resolution cryo-EM structures. This work is followed by operations related to cryo-EM SPA, including cryo-EM sample preparation and vitrification, EM imaging, data collection and processing, 3D model reconstruction, and refinement.
We have applied this technique to the target identification and hit identification phases of the drug discovery process and are ready to push it to the lead compound optimization cycle. Creative Biostructure focuses on target protein structures most concerned by the pharmaceutical industry, such as GRCR, ion channels, kinases, and some biological macromolecular complexes. Moreover, we have extensive experience in using cryo-EM SPA to obtain structural information of membrane proteins, large protein complexes and weakly expressing targets, and to perform epitope mapping of antibody-antigen interactions.
In structural biology, NMR spectroscopy is a complementary approach for X-ray crystallography to determine the structure of biological macromolecules, especially the structural analysis of peptides and low-molecular-weight proteins. For some proteins that are difficult to obtain single crystals due to their high flexibility, high-resolution structures can be obtained by generating high-quality NMR spectra. In addition to structural information, NMR spectroscopy can also provide dynamics information and information on protein interaction interfaces.
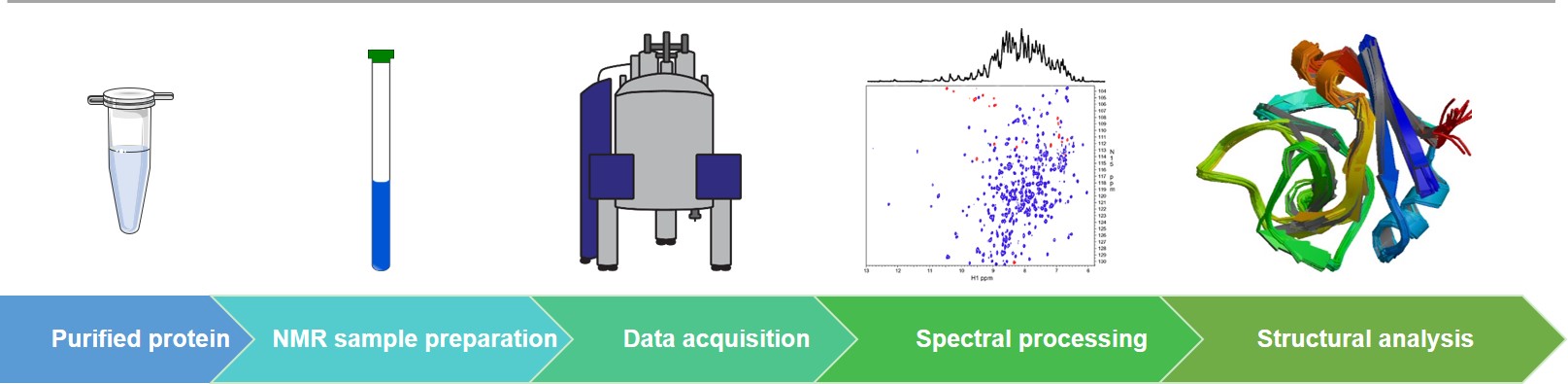
Over the past decade, backed by the advanced NMR spectroscopy platform, Creative Biostructure has solved many high-quality 3D biomacromolecule structures, including nucleic acids and proteins. Nowadays, we apply our rich expertise and experience in NMR spectroscopy to the drug discovery process. Our extensive and customized protein NMR spectroscopy services include, but are not limited to:
Our protein NMR spectroscopy service covers the four main steps of NMR experiments, namely sample preparation, data collection, spectral processing, and structural analysis. We can perform multidimensional (1H/1H, 1H/15N, or 1H/13C) NMR experiments as the basis for detailed structural analysis. The target protein samples for analysis can be produced by us or obtained from customers. For larger proteins, we support the production of isotope-labeled samples and conduct 3D NMR experiments.

Easy access to products and services you need from our library via powerful searching tools